- Home
- Weddings
- Portraits
- Journal
- Contact
- Danganronpa characters v2
- Star trek adventures form fillable character sheet
- Toshiba r830 battery life
- In the house in a heartbeat 28 days later resident evil mashup
- D2 lod runewords 1-14
- Airpods down by marian hill
- Japan exclusive wii channels
- Wondershare quiz creator 4-5-1 full
- Command and conquer red alert 3 uprising compatibility windows 10
- Free hyperterminal
- Trucos para ultimate spiderman pc
- Which f1 pc game is the best version
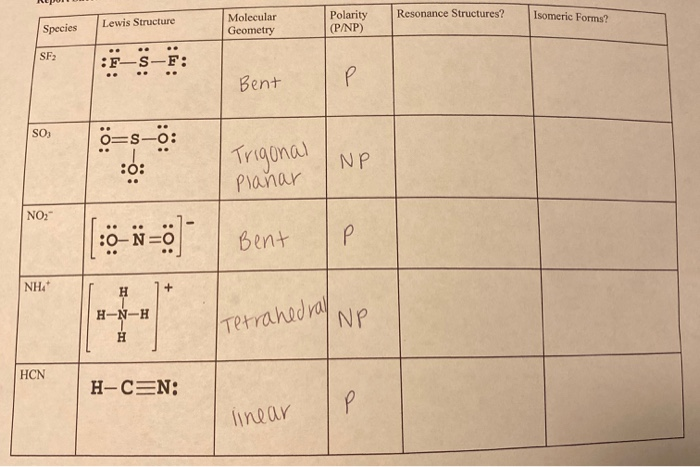
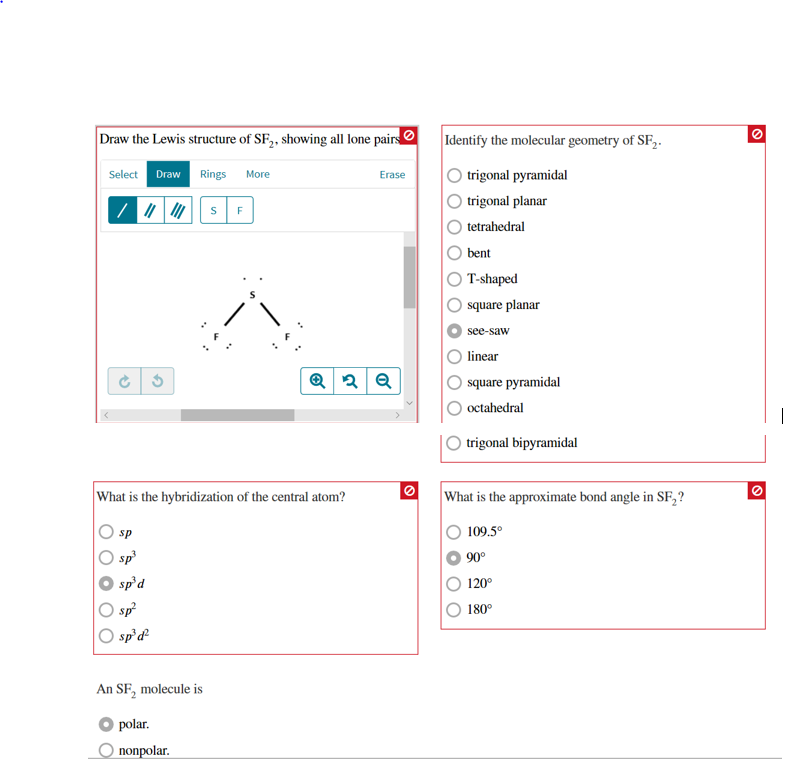
- Sf2 lewis structure molecular geometry
- Playboy magazine wiki
- Hp laptop coprocessor missing
- Adobe photoshop cc 2017 torrent
It is very helpful to learn the hybridization and VSEPR theories at his point which suggest that the hydrogens and the lone pairs on the oxygen are in sp 3 orbitals at ~109. Dipole moments can “cancel”, giving a net non-polar molecule. A positive value means that the induced dipole moment points from the metal to MoSSe.In a similar manner the configurations of methane (CH 4) and carbon dioxide (CO 2) may be deduced from their zero molecular dipole moments. The experiments were performed on the qR(0)00 and the q Vapour spectra and solution spectra in a variety of solvents were recorded for some D,D′ Ar A and A,A′ Ar D molecules, where D D′ NH2 and A A′ NO2. Dipole moment ($\mu $) = Charge (Q) x distance of separation (r) The dipole moment is usually expressed in the Debye unit (D). NO2+ has the largest angle, followed by NO2, and NO2 moment. Phosphorus is negatively polarized and hydrogen is positively polarized.No2 dipole moment Rotational excitation cross section. But, due to electronegativity difference, phosphorus and hydrogen atoms are slightly polarized. There are no direct charges such as -1,+1 on atoms. Are there charges on atoms in PH 3 lewis structure? So, shape of both ammonia and phosphine is trigonal pyramidal. Therefore geometry should be tetrahedral.Īsk your chemistry questions and find the answers What are the similarities in lewis structures of ammonia and phosphine?īoth structures have three sigma bonds and one lone pair on center atom. Summation of number of sigma bonds and lone-pairs around phosphorus atom is four. Therefore, shape of PH 3 is trigonal pyramidal. There are three sigma bonds and one lone-pair around phosphorus atom. So, we have got the best lewis structure for PH 3. Check the stability and minimize charges on atoms by converting lone pairs to bondsīecause there is no charges on atoms, no need to reduce charges as a step of obtaining the best lewis structure. Also, remember that PH 3 is a molecule which does not have a There are no charges on phosphorus atom and hydrogen atoms. Therefore, then mark that electron pair on center atom phosphorus.Therefore, we cannot mark that electrons pair on hydrogen atoms. But in PH 3, hydrogenĪtom are the outside atoms which cannot keep more than two electron in its last shell. Usually, those remaining electron pairs should be started to mark on outside atoms.There are already three P-H bonds in the above drawn sketch.Remember that, there are total of four electron pairs. Mark lone pairs on atomsĪfter determining the center atom and sketch of PH 3 molecule, we can start to mark lone pairs on atoms. Now, we can draw the sketch of PH 3 to describe how atoms are attached with each other. Therefore, phosphrus atom should be the center atom of Phosphorus, which atom has the highest valence? Maximum valence
To be the center atom, ability of having greater valance is important. Total electron pairs are determined by dividing the number total valence electrons by two. Pairs = σ bonds + π bonds + lone pairs at valence shells

HydrogenĮlectron in its last shell (valence shell). There are two elements in PH 3 hydrogen and phosphorous. Total number of electrons of the valance shells of PH 3 Check the stability and minimize charges on atoms by converting lone pairs to bonds to obtain best.Mark charges on atoms if there are charges on atoms.Total electrons pairs as lone pairs and bonds.Find total number of electrons of the valance shells of hydrogen atoms and phosphorous atom.But, because phosphine is a simple molecule, these steps are notĬomplex and do not require all general steps which are used to draw lewis structures of complex molecules and ions. There are several steps to draw the lewis structure of PH 3. Therefore, we can draw the lewis structure According to the lewis structure shown above, you will understand phosphine's structure is a simple.
